Genomic Biomarkers and Clinical Perspectives of Fluoropyrimidines in Cancer Treatment. A Literature Review
DOI:
https://doi.org/10.4238/p6r02x72Keywords:
Fluoropyrimidines, cancer, dihydropyrimidine dehydrogenase, gene variant, toxicityAbstract
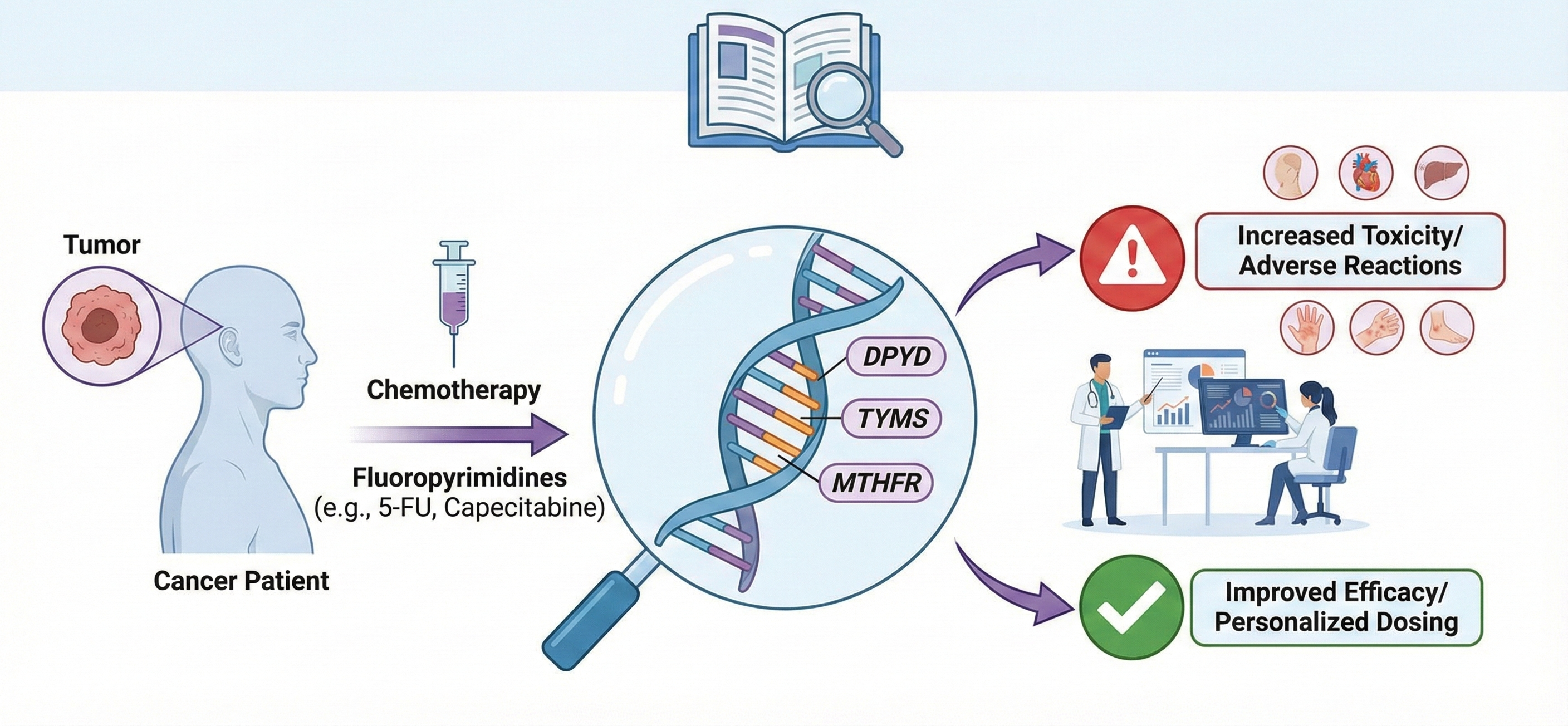
Fluoropyrimidines, such as 5-fluorouracil (5-FU), are vital chemotherapy agents against gastric, colorectal, and breast cancer. Despite their efficacy, response and toxicity vary greatly between patients, prompting the search for genomic biomarkers to personalize treatment. In gastric cancer, combinations with fluoropyrimidines and oxaliplatin improve survival, while in colorectal cancer, regimens with 5-FU, oxaliplatin, and irinotecan achieve a 3-year survival rate of 86%, albeit with considerable toxicity. In breast cancer, capecitabine is effective in anthracycline-resistant cases but requires careful risk-benefit assessment. The metabolism of 5-FU is key to understanding these variations. The enzyme dihydropyrimidine dehydrogenase (DPD), encoded by DPYD gene, catabolizes 5-FU. Genetic variants such as DPYD2A (c.1905+1G>A) and c.2846A>T are associated with DPD deficiency, increasing the risk of severe toxicity and mortality. Detection of these variants or elevated blood uracil levels (uracilemia >16 ng/mL) allows for dose adjustments or treatment contraindications. Thymidylate synthase (TS), the target of 5-FU, also influences resistance. Variants in its regulatory region (2R/3R) modulate its expression, affecting efficacy in colorectal and gastric cancer. Other biomarkers include variants in MTHFR, which impact folate availability, and the expression of orotate phosphoribosyltransferase (OPRT) and transporters such as ABCC5. Pharmacogenomics is essential to optimize the safety and efficacy of fluoropyrimidines. Prior genetic screening for critical variants, dose adjustments, and plasma monitoring are recommended. Validation and standardization of these biomarkers across diverse populations is crucial for advancing precision oncology that improves survival and reduces toxicity.
Downloads
Published
Issue
Section
License
Copyright (c) 2025 Diego Saldaña, Karol Montealegre, Carlos Castro, Mauricio Rey (Author)

This work is licensed under a Creative Commons Attribution-ShareAlike 4.0 International License.



